 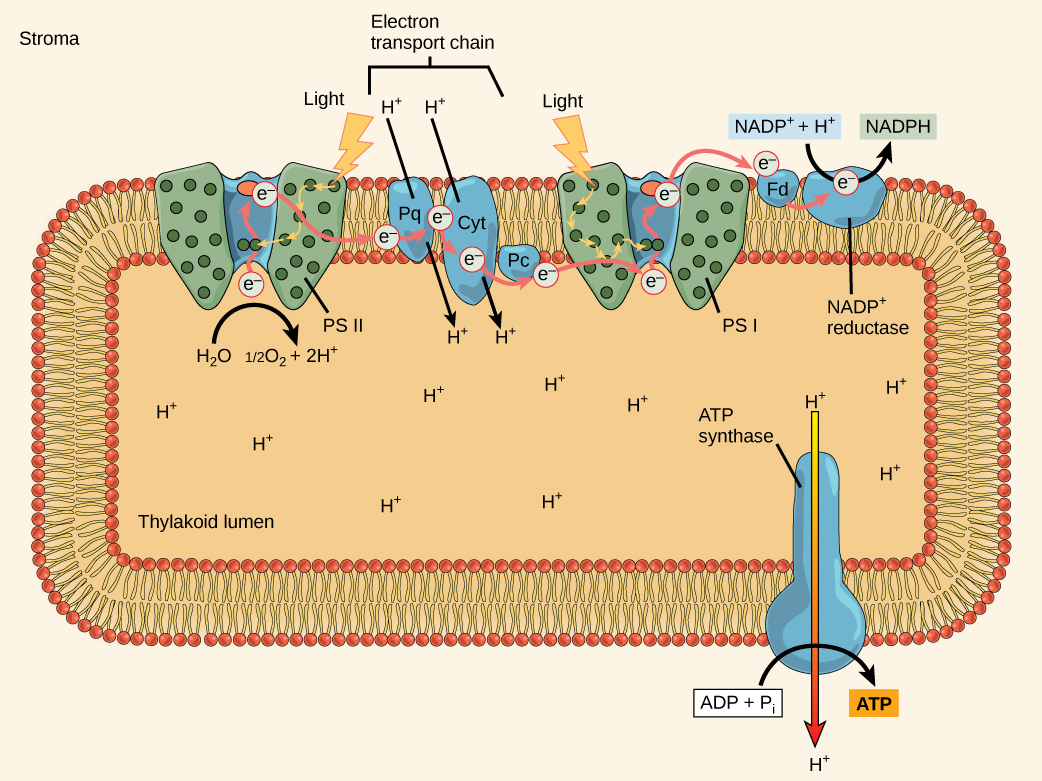
Again, this supplies energy for ATP synthesis. 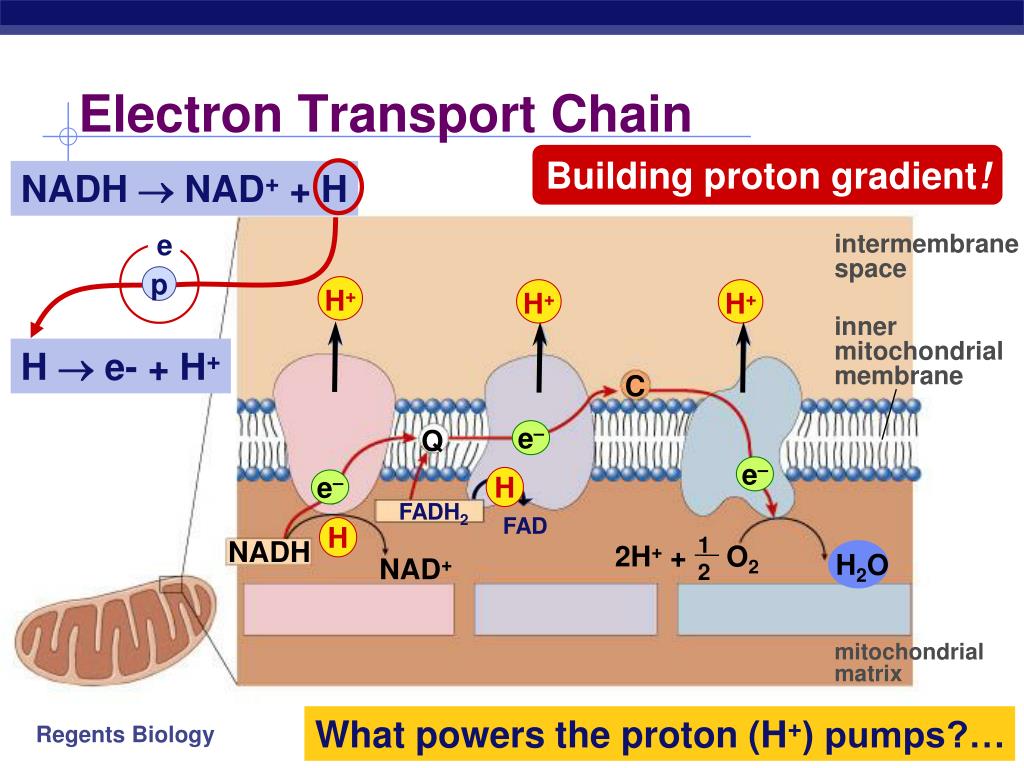
Electron flow through Complex II transfers proton(s) through the membrane into the intermembrane space. It accepts electrons from reduced coenzyme Q, moves them within the complex through two cytochromes b, an iron‐sulfur protein, and cytochrome c 1. Coenzyme Q is not bound to a protein instead it is a mobile electron carrier and can float within the inner membrane, where it can transfer electrons from Complex I and Complex II to Complex III.Ĭomplex III is also known as coenzyme Q‐cytochrome c reductase. Figure 2 shows the quinone, semiquinone, and hydroquinone forms of the coenzyme. The reduction potential of FAD is ‐0.22 V, as opposed to ‐0.32 V for NAD.Ĭoenzyme Q is capable of accepting either one or two electrons to form either a semiquinone or hydroquinone form. This experimental observation also fits with the difference in the standard reduction potentials of the two molecules. Because translocated protons are the source of the energy for ATP synthesis, this means that the oxidation of a molecule of FADH 2 inherently leads to less ATP synthesized than does the oxidation of a molecule of NADH. No protons are translocated by Complex II. Electrons flow from succinate to FAD (the flavin‐adenine dinucleotide) coenzyme, through an iron‐sulfur protein and a cytochrome b 550 protein (the number refers to the wavelength where the protein absorbs), and to coenzyme Q. 
Apparently, conformational changes in the proteins of Complex I also are involved in the mechanism of proton translocation during electron transport.Ĭomplex II, also known as succinate‐coenzyme Q reductase, accepts electrons from succinate formed during the TCA cycle. One possible source of the protons is the release of a proton from NADH as it is oxidized to NAD, although this is not the only explanation. Complex I is capable of transferring protons from the matrix to the intermembrane space while undergoing these redox cycles. First, the flavin coenzyme (flavin mononucleotide) and then the iron‐sulfur center undergo cycles of reduction and then oxidation, transferring their electrons to a quinone molecule, coenzyme Q(see Figure 1). The electrons flow through a flavoprotein containing FMN and an iron‐sulfur protein. The NADH releases a proton and two electrons. Complex I, also called NADH‐coenzyme Q reductase, accepts electrons from NADH. There are four major protein‐membrane complexes.Ĭomplex I and Complex II direct electrons to coenzyme Q. The inner membrane of the mitochondrion is tightly packed with cytochromes and proteins capable of undergoing redox changes. Peter Mitchell, a British biochemist, essentially by himself and in the face of contrary opinion, proposed that the mechanism for ATP synthesis involved the coupling between chemical energy (ATP) and osmotic potential (a higher concentration of protons in the intermembrane space than in the matrix). This model for ATP synthesis is called the chemiosmotic mechanism, or Mitchell hypothesis. Oxidative Phosphorylation: Energy YieldsĮlectrons flow through the electron transport chain to molecular oxygen during this flow, protons are moved across the inner membrane from the matrix to the intermembrane space.Metabolism: A Collection of Linked Oxidation and Reduction Processes.Biosynthetic versus Catabolic Reactions.Chemical Mechanisms of Enzyme Catalysis.Physiological Conditions and Hemoglobin.Oxygen Binding by Myoglobin and Hemoglobin.Overview of Biological Information Flow.Water: Properties and Biomolecular Structure.United Strength of Biochemical Structures.Electrostatic and van der Waals Interactions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
